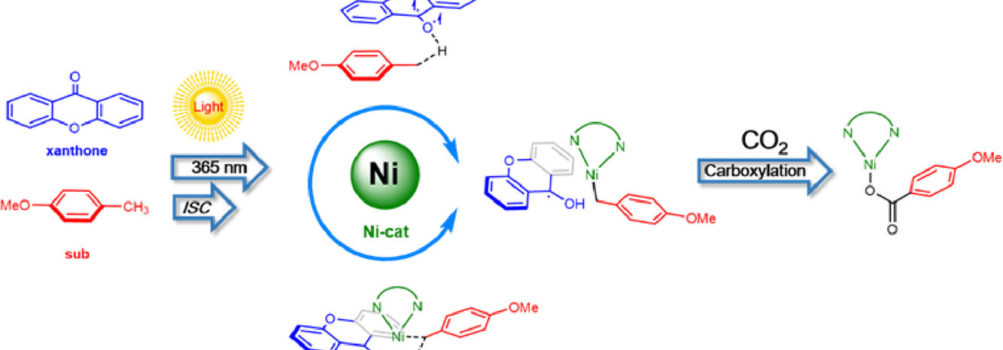
NordCO2 PI Mårten S. G. Ahlquist and collaborators have explored the mechanism behind the photo carboxylation of C(sp3)-H bonds in benzylic substrates using DFT calculations. To use their own words, “The present study sheds light on the detailed functions of the photo-catalyst and transition-metal catalyst in the carboxylation reactions, and the interactions might be possibly generalized to other photoredox systems with co-catalysts“. Congratulations to all authors!

Title:
Mechanistic study on the photo carboxylation of benzylic C-H bonds by xanthone and Ni(0) catalysts
Abstract:
The photo carboxylation of the benzylic C(sp3)-H bond catalyzed by xanthone/nickel were examined by density functional theory (DFT) and time-dependent DFT (TD-DFT) calculations. This study corroborates the previous proposal that light promotes the H-transfer from benzylic C(sp3)-H bond of the p-methoxytoluene to excited state of photocatalyst xanthone. Meanwhile, Ni(0) catalyst could mediate the H-transfer to occur via an electron-coupled-proton transfer manner, and then remarkably facilitates the carboxylation step (compared to the Ni-absent systems). After that, the generated Ni(I) intermediate and ketyl radical anion complete the carboxylation and electron transfer processes independently.
You can find a list of publications by NordCO2 on our Publications page.