Title:
Efficient palladium-catalyzed electrocarboxylation enables late-stage carbon isotope labelling
Abstract:
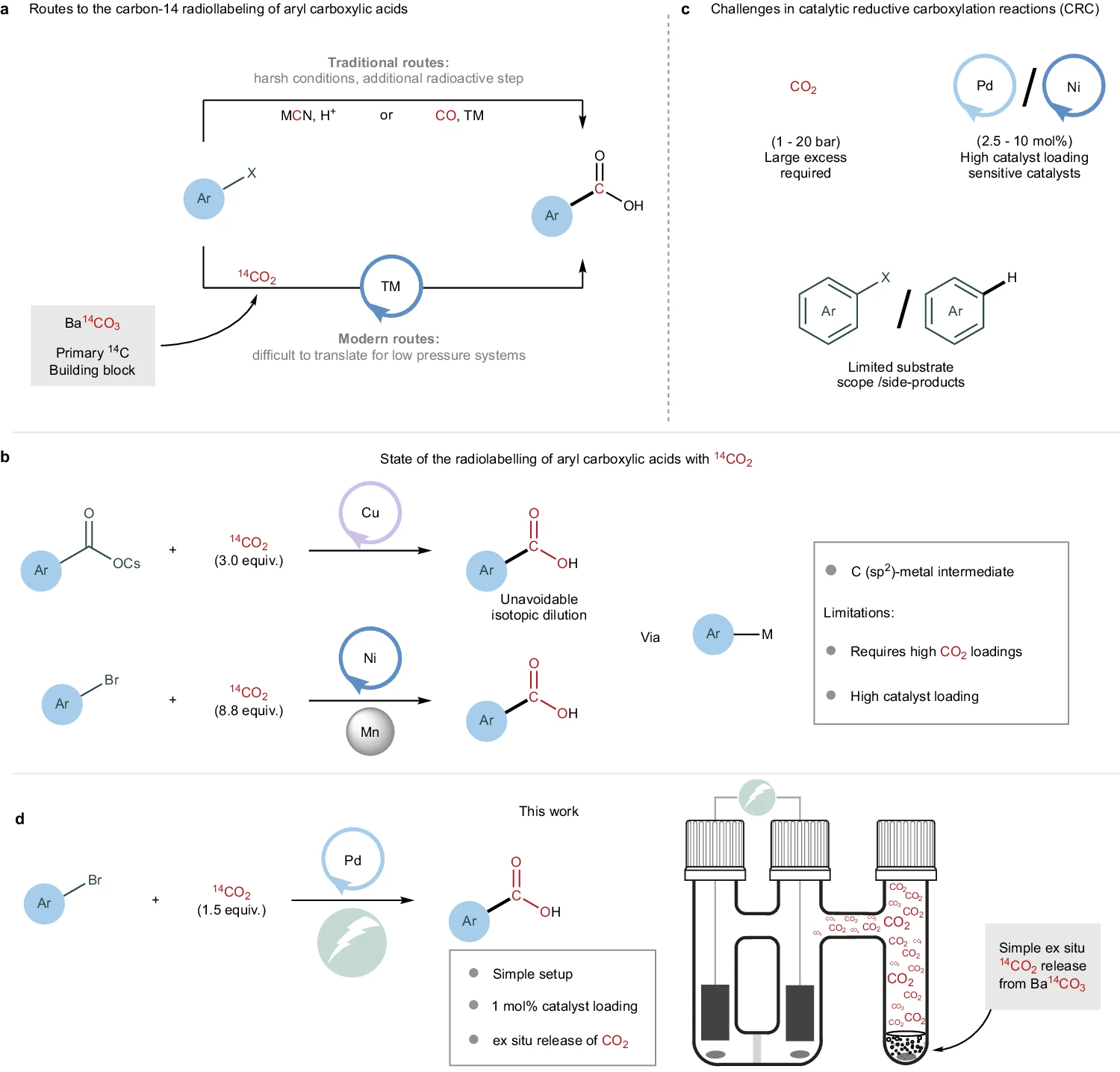
Carbon isotope labelling of bioactive molecules is essential for accessing the pharmacokinetic and pharmacodynamic properties of new drug entities. Aryl carboxylic acids represent an important class of structural motifs ubiquitous in pharmaceutically active molecules and are ideal targets for the installation of a radioactive tag employing isotopically labelled CO2. However, direct isotope incorporation via the reported catalytic reductive carboxylation (CRC) of aryl electrophiles relies on excess CO2, which is incompatible with carbon-14 isotope incorporation. Furthermore, the application of some CRC reactions for late-stage carboxylation is limited because of the low tolerance of molecular complexity by the catalysts. Herein, we report the development of a practical and affordable Pd-catalysed electrocarboxylation setup. This approach enables the use of near-stoichiometric 14CO2 generated from the primary carbon-14 source Ba14CO3, facilitating late-stage and single-step carbon-14 labelling of pharmaceuticals and representative precursors. The proposed isotope-labelling protocol holds significant promise for immediate impact on drug development programmes.
Find a list of publications by NordCO2 members on our Publications page 🙂